Prof. Dr. Wolfgang Voelter Laboratories Complex (NRL)

A research wing of the H.E.J. Research Institute of Chemistry is focusing on applied chemical science, textile chemistry, nanochemistry and other disciplines of national need. The Executive Board of ICCBS approved that the New Research Laboratory (NRL) of the ICCBS will be named as the Prof. Wolfgang Voelter Laboratories Complex, in recognition of his outstanding services to the ICCBS and Pakistan, spread over 5 decades.
Prof. Dr. Wolfgang Voelter is among those individuals who have left an indelible mark on science in Pakistan and in many other developing countries. He was born in October 20, 1936 in Ludwigsburg, Germany, and completed Diploma in Chemistry in 1956 from Tübingen University. He also got degree in Vorphysikum in Medicine (1964) and Dr. rer. nat. (1966) from Tübingen University (Prof. Ernst Bayer). In parallel, he finished Physikum in Medicine at the Erlangen University. He has worked as a Research Fellow at Stanford University (Prof. Carl Djerassi) and Kaiser Foundation Research Institute, California, USA, from 1966-1970. Since 1970 he is associated with Tübingen University as a Professor.
He first visited Pakistan in September 1974, and met Professors Salimuzaman Siddiqui FRS and Atta-ur-Rahman FRS at the Department of Chemistry, University of Karachi. He convinced German Government and GTZ to approve an initial allocation of 2.3 million DM, followed by another allocation of 3.5 million DM. This allowed H. E.J. Research Institute of Chemistry, to acquire sophisticated equipment, such as mass (MS), nuclear magnetic resonance (NMR), circular dichorism (CD) instruments, and further laboratory equipment, chemicals, and text books.
Prof. Voelter has trained and supported a large number of young Pakistanis in their careers as a true mentor. Approximately 40 Pakistani scientists, students, and technicians have got advance research training in the esteemed laboratory of Prof. Wolfgang Voelter, including 20 full Ph.Ds. Over 90 German students from his laboratory have also visited the HEJRIC to carry out part of their Diploma work. Over 150 research publications in best peer reviewed journals have been published in collaboration with the laboratory of Prof. Wolfgang Voelter.
Prof. Voelter has received several international awards, including prestigious civil awards, Hilal-e-Pakistan, and Sitara-e-Pakistan, from the Government of Pakistan. He has an unparalleled love, affection, and respect for the people of Pakistan, and confidence on their talent and abilities. He is truly the ambassador of Pak-German Scientific cooperation and friendship between the two great nations.
Core Facility:
Core Facility:
 NMR Facility |
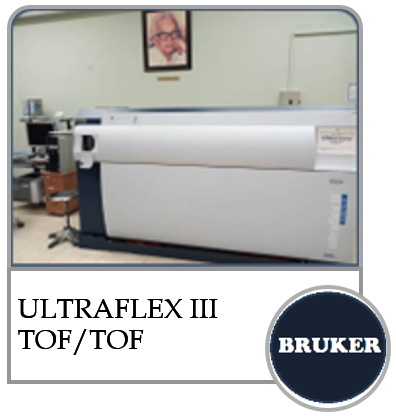
 Mass Spectroscopy |
.jpg) Single Crystal X-Ray Diffraction Facility |
 Analytical Laboratory |
NMR Spectroscopy Core Facility

Mass Spectroscopy

Single-Crystal X-Ray Diffraction Facility

Analytical Laboratory












.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)